EMAUD 2023
European Market Access Diploma
Gain key insights into the market access governing global healthcare and explore how effective policy can be used to address contemporary challenges.
REGISTRATION FOR NEXT MODULE
COMMING SOON
Duration
Time commitment
Language
Video transcript
Difficulty
Platform
Who can benefit from this course?
Market Access Experts
Individuals who are already established in the field of market access, but who are seeking to further refine their understanding and solidify their existing knowledge base.
Non-MA Industry Associates
Individuals who are already established in the field of market access, but who are seeking to further refine their understanding and solidify their existing knowledge base.
Market Access Transitioners
Pharmaceutical or consulting industry professionals aiming to transition into market access roles.
Students
Students and individuals who are just embarking on their professional journeys and who are interested in exploring the exciting field of market access
Why apply?
increase of the interest of employers in course participants in recent years, according to LinkedIn
job offers in the sector require the knowledge contained in the course
increase of the interest of employers in course participants in recent years, according to LinkedIn
What you'll learn?
- Set up a successful market access strategy.
- Develop, validate and execute a market access plan.
- Build value story to optimize market access.
- Understand strengths and pitfalls of available evidence.
- Design pricing research.
- Anticipate the future paradigm changes in market access.
- Acquire le market access mindset.
Course description
Market Access is a complex process. It is often referred to as the fourth hurdle in drug development and has become an inescapable reality today. It is nowadays the driver of the global income of a new product/drug. No company providing drugs or devices cannot expect to succeed without designing a relevant market access strategy early in the development process, and an effective execution planning.
The concept of Market Access requires as much knowledge as professional capabilities. The EMAUD program is situated at a crossroads of multiple disciplines that all form an integral part of a valid comprehensive course.
Duration
Time commitment
Language
Video transcript
Difficulty
Platform
Ours instructors
Lylia Chachoua
Senior Consultant PRMA in Clever-Access
Hervé Lamarque
VP at Incyte Head of EU Access, Pricing, HEOR and Business development
Isaac Odeyemi
Head of Outcomes Research
Wael Belgaied
Pricing & Market Access Consultant in Inov Intell
Emilie Clay
Co-Founder of Clever-Access
Vivian Chen
Professor and Senior Researcher at Tsinghua University-Hospital Management Institute
Philippe Larame
Head of Payer Engagement, Capabilities & Innovation at Ipsen Headquarters
Ulf Person
Senior Advisor at IHE - The Swedish Institute for Health Economics
Keith Tolley
Director at Tolley Health Economics
Ataru Igarashi
Associate Professor at Yokohama City University School of Medicine
James Chambers
associate professor at Tufts Medical Center Institute for Clinical Research and Health Policy Studies
Dávid Danko
managing partner at Ideas & Solutions (I&S)
Emaud has a global outreach. Our students come from
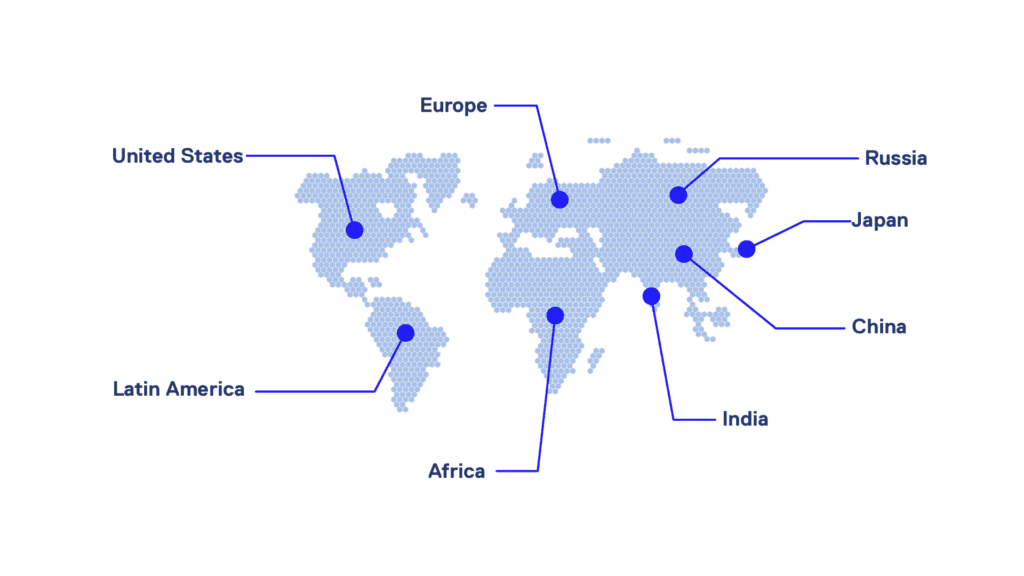
of places are granted with waivers from 100% to 30% to people from developing countries, people off job, or people with low salary in the life science field who wanted to access to a new career.
What do you need to know ?
How many modules are included in the course?
The program consists of 5 mandatory modules plus one optional module (3 days) on Health Economics. The 5 modules can be done separately i.e., not necessarily in only one year, and not in any specific order.
Practical or theoretical?
- Each module will contain one case study and one workshop.
- Practical applications for each main topic.
- Multiple examples from the public domain and lecturers experience.
- The full development of a Market Access Plan including strategy, gap analysis, activity prioritization.
Where can I find the exact outline of the course modules?
You can find it here
Do I have to submit the thesis at the end?
- The main objective of the thesis is to demonstrate your ability to analyze and think strategically in relation to any market access question.
- Your thesis can cover one or several of the following topics: Literature review conducted according to state of art, Review of HTA or pricing decisions, Landscape analysis, Case Study, Health economics project, Pharmaco-epidemiology project, Pricing research, Market Access Strategy, Market Access perspectives and drug development, HEOR applied to market access, Etc.
- The thesis should be in English and should not exceed 50 pages or a maximum of 50 PowerPoint slides. There are no minimum slides or pages. What count is value not quantity.
- It could eventually be done jointly with up to 3 candidates maximum.
- For sensitive case studies, we exert full confidentiality, ensuring that the thesis is not available to any third party and stored safely. A confidentiality agreement will be signed, and the thesis returned to the candidate. However, when possible, we prefer to avoid confidential topics.
- On successful completion of your course and positive evaluation of your research work, you will receive a diploma delivered by MAS. We do not deliver grade but positive or negative decision.
Do I have to meet any requirements?
Students currently undertaking a Master’s I or equivalent in Medicine, Pharmacy, Dentistry (all under 5 years);
Professionals who have experience working in market access. Other candidates with equivalent qualifications willing to undertake a career in market access.
What is the exact date of the course?
The exact dates of the modules in 2023 are:
Module 1: 16.10.2023 – 20.10.2023
Module 2: 11.12.2023 – 16.12.2023
Module 3: 05.02.2024 – 09.02.2024
Module 4: 08.04.2024 – 12.04.2024
Module 4 bis: 20.05.2024 – 23.05.2024
Module 5: 17.06.2024 – 21.06.2024
How can I apply?
Please send CV and a cover letter to: administration@emaud.eu
Once received, your application will be reviewed by a selection committee. We will then revert to you within two weeks to give you the committee’s decision.
How much does the course cost?
€8,000 diploma
€1,600 per module
*) The attendance to EMAUD courses is compulsory. Tuitions and scholarship fees will not be refunded in case of absence.
Employees of these companies participated in the course.